Advancing Functional Connectomics with Bidirectional, Non-Invasive Magnetogenetic Tools
The quest for a truly non-invasive, precise tool to regulate neuronal circuits—the field known as neuromodulation—has taken a significant step forward. A groundbreaking study recently published in Neuroscience (PMCID: PMC10369996) has reported a novel suite of magnetogenetic tools capable of regulating neuronal activity when exposed to magnetic fields. Unlike traditional optogenetics, which requires a surgically implanted fiber optic device, this system allows for highly cell-specific, temporally precise, remote, and reversible modulation of deep brain structures in awake, freely moving mice.
The core of this technology is a single anti-ferritin nanobody fused to a TRPV1 receptor. This construct is remarkably efficient because it relies on endogenous mammalian channels and tethers to endogenous ferritin, bypassing the need for exogenous synthetic magnetic nanoparticles that can cause off-target activation. Furthermore, the entire cassette can be packaged into a single AAV vector, simplifying delivery and ensuring consistent dosing.
Excitatory Regulation and Motor Circuit Activation
When researchers delivered a floxed nanobody-TRPV1Ca2+ construct into the striatum of adenosine 2a receptor-cre driver mice via AAV, the animals exhibited robust motor freezing when placed in a 3T MRI or adjacent to a transcranial magnetic stimulation (TMS) device. The team determined that a magnetic field threshold of 180 mT was sufficient to induce these behavioral alterations. To validate the underlying cellular response, neuronal activation was confirmed in vivo using immediate early gene c-fos expression, 18F-FDG PET imaging, and fiber photometry.
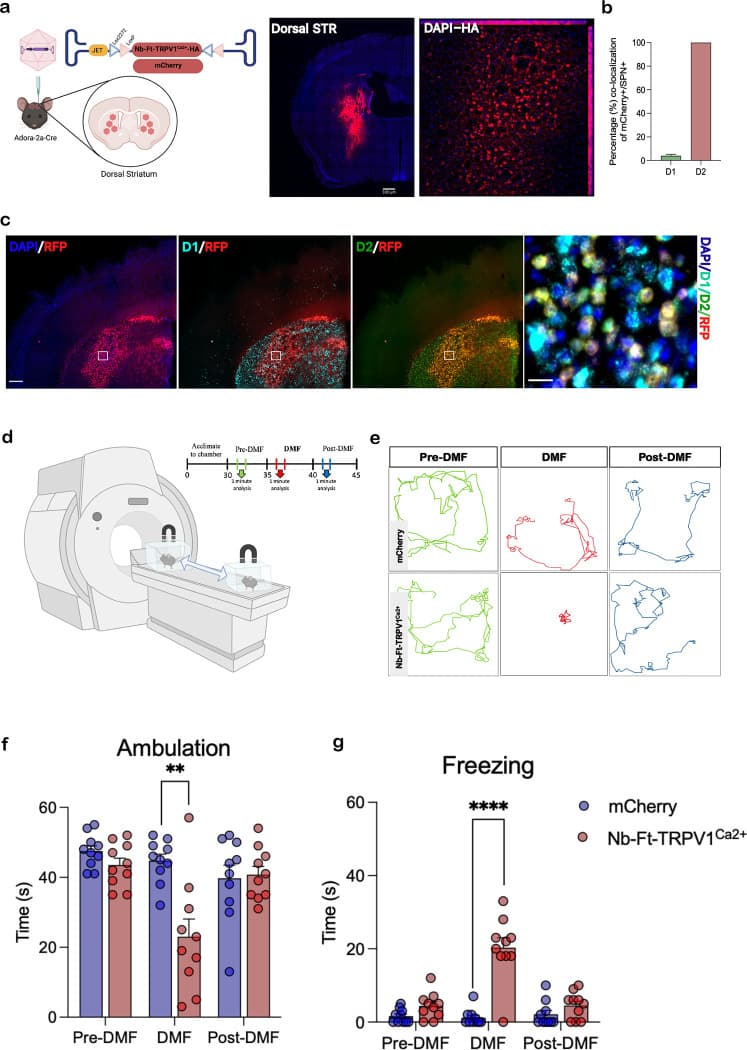
Inhibitory Regulation for Parkinsonian Models
To achieve bidirectional control, the research team generated a mutation to gate chloride ions, creating an inhibitory construct known as Nb-Ft-TRPV1Cl-. They applied this inhibitory magnetogenetic system to a Parkinson disease model by targeting the subthalamic nucleus in PitX2-cre hemiparkinsonian mice. Application of a magnetic field significantly reduced local c-fos expression and led to a marked reduction in apomorphine-induced motor rotational behavior.
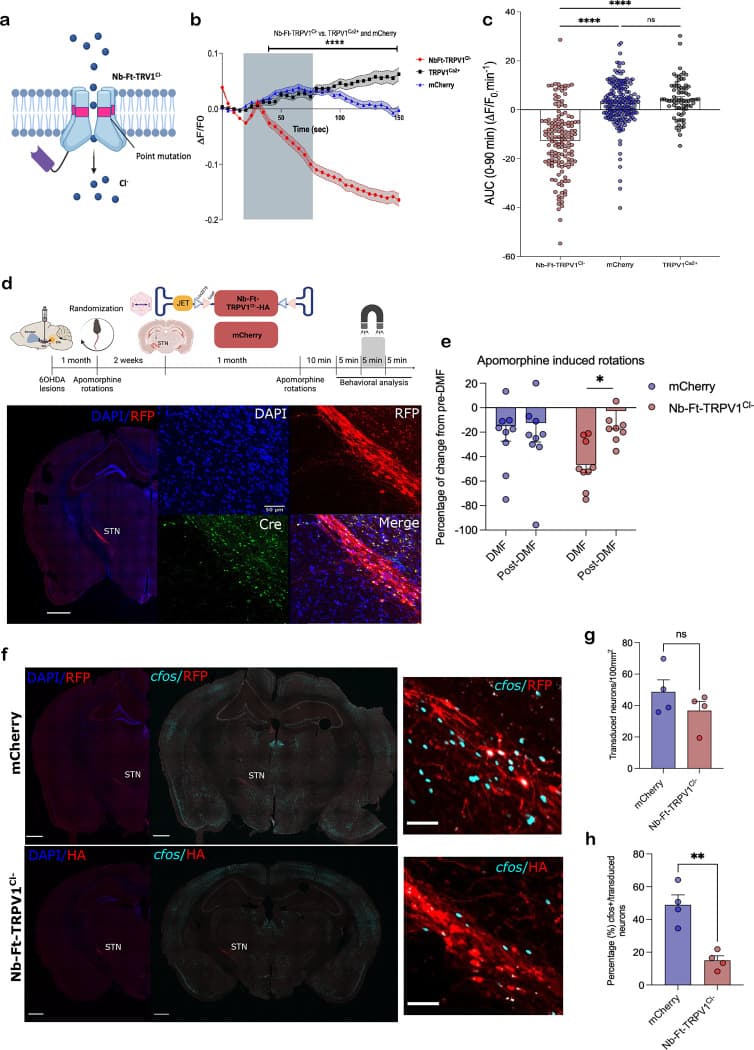
Translational Potential for Pronewbio
Our company, a leader in providing high-performance viral vector solutions, is proud to fully support researchers in adopting this cutting-edge technology. Building directly on the methodology described in PMC10369996, we now offer optimized Nb-Ft-TRPV1 viral vector products (AAV-hSyn-Nb-Ft-TRPV1 family) that are ready for immediate use. Whether your studies demand precise neuronal excitation or silencing, our customizable tools, backed by extensive quality control, can be tailored to meet your specific protocol requirements and large-batch needs. We are dedicated to empowering your research in the fast-growing field of non-invasive connectomics.
