Large-scale Recombinant Antibody Production Services
Moving a therapeutic antibody from early-stage discovery into preclinical trials requires a dependable manufacturing partner. The transition from bench-scale testing to bulk production is often fraught with challenges, including decreased yields, aggregation, and altered protein behavior.
At Pronewbio, we specialize in overcoming these scale-up hurdles. Our large-scale recombinant antibody production services are designed to provide a continuous, high-quality supply of your therapeutic candidates, delivering everything from multi-gram to kilogram quantities tailored to your exact project timeline.
Why Partner with Pronewbio for Scale-Up?
- Seamless Bench-to-Bulk Scale-Up: We know that scaling up is not simply about increasing bioreactor volume. Our engineering team meticulously optimizes expression conditions during the pilot phase, ensuring a smooth and predictable transition from small-scale milligram batches to large-scale kilogram manufacturing without compromising molecular integrity.
- Optimized Expression Platforms: We utilize industry-standard, high-density HEK293 and CHO suspension cell lines. Whether your pipeline requires the rapid turnaround of transient expression or the robust yield of stable pool generation, our platforms are highly adaptable to various antibody formats, including full-length IgGs, scFvs, and VHHs.
- Unwavering Quality Consistency: Batch-to-batch consistency is the cornerstone of reliable in vivo and toxicology studies. Through rigorous downstream purification and stringent quality control protocols, we guarantee that your scaled-up bulk antibodies perfectly match the biophysical properties, purity, and efficacy of your initial small-scale prototypes.
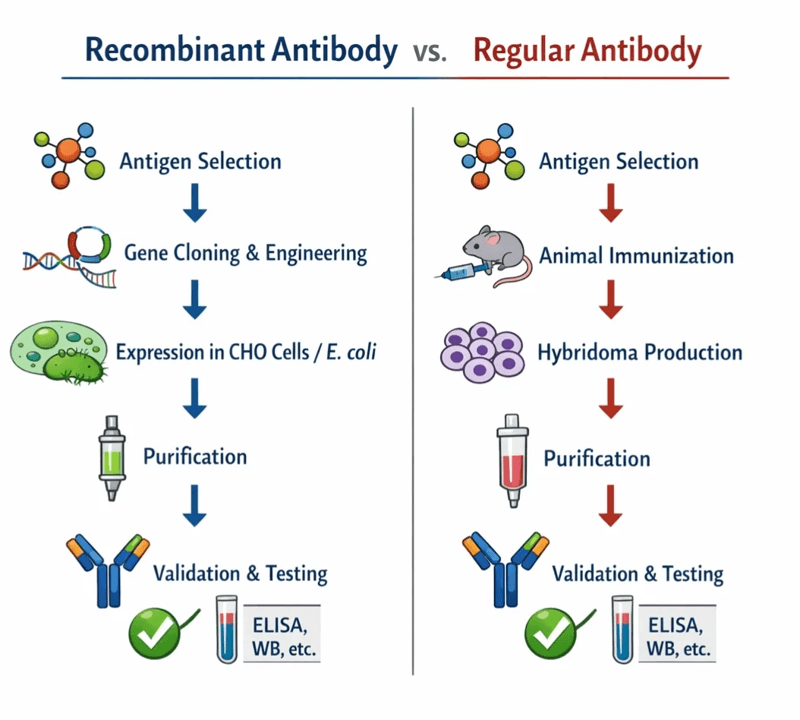
Our Streamlined Production Process
- Gene Synthesis & Vector Construction: Rapid sequence optimization and cloning into our proprietary high-yield vectors.
- Pilot Expression & Optimization: Small-scale testing to establish optimal feeding strategies and predict large-scale behavior.
- Large-Scale Cell Culture: Volume expansion in advanced bioreactors, carefully monitored for optimal cell viability and productivity.
- Purification & Quality Control: Multi-step chromatographic polishing followed by comprehensive QC (including SEC-HPLC, SDS-PAGE, and ultra-low endotoxin testing) to ensure absolute safety and purity.
Case Study
Production of Anti-Amyloid Beta 1-5 Antibody, Clone 3D6 (Mouse IgG1 kappa)


Secure Your Therapeutic Supply Chain
Advancing a therapeutic candidate from discovery to preclinical validation requires more than just raw manufacturing capacity; it demands a strategic partner who understands the biophysical intricacies of your specific molecule. Let Pronewbio de-risk your scale-up journey with our proven bioprocessing expertise, rigorous quality systems, and unwavering commitment to your project timeline.
Ready to Scale Your Production? Consult with our biomanufacturing scientists today to discuss your specific yield requirements, evaluate cell line compatibility, and receive a customized, transparent project proposal.
